Project planning
The process of developing a guideline can be long and complex — it needs good organisation and planning to be successful
Project planning
A guideline typically takes between 18–30 months to complete, with reported costs of up to approximately AUD $3 million for major national guidelines.* While work is underway on how to accelerate the production of guidelines, the reality is that the fundamental processes of guideline development such as prioritisation of outcomes, the evidence review, developing the recommendations and consultation take time, careful planning and adequate resourcing.
As with any complex project, planning needs to be done before work on a guideline begins, and typically this is done by an organising team or steering committee. Guideline planning includes sourcing and allocating funding, establishing project governance, establishing timelines, scoping the topic and planning the methods to be used. You need to establish the guideline development group and determine the roles and tasks of the various people you bring on board. You also need to consider who else needs to be involved, how they should be involved and when, for example, consumers, members of community groups and other stakeholders. Details of all processes and procedures used to develop the guideline (Standard 2.1) should also be made available as part of the administrative report of the guideline.
The end product will be a project plan that can be updated as the project progresses. It should include a budget with the estimated costs for each of the development group processes, such as holding meetings and hiring experts to evaluate the evidence and dissemination products. The project plan should cover the different phases of the project and identify any contingencies and redundancies.
This module provides practical advice on the organisation, planning and budgeting of a guideline. It includes a broad overview of the guideline development process, some suggested planning approaches and other issues to consider. It includes some suggested items required to develop a guideline that should be included in the budget.
The steps in this module are not intended to be prescriptive. Depending on your situation some steps may need to be considered before others — budgeting, for example. Other modules that may be helpful at this stage include Guideline development group, Consumer involvement, Training, Adopt, adapt or start from scratch, and Identifying and managing conflicts of interest.
What to do
1. Consider the need for your guideline
In the early stages of guideline planning prepare to justify the need for your guideline. Guideline development is a long and expensive undertaking. Organisations — including your own — that will be funding and approving your guideline will need some evidence that the guideline will be relevant and useful before committing to the project. You should prepare a ‘business case’ for the guideline that can be used in these discussions which may include questions like:
- Is this guideline really needed? Are there alternatives?
- What is the purpose of your guideline?
- How will this guideline impact the current burden of disease, including health system costs?
- How will this guideline address the key priority areas on your topic and the contextual issues important to these areas? (see the Scoping the guideline module)
- Will it still be needed in two to three years’ time?
- Are there existing national and/or international guidelines on this topic, and if so, how will yours be different?
- What resources will be needed and how long will the process take?
- Will your guideline be feasible to implement (see the Implementability module).
Chapter 2 of the WHO Guideline Development Handbook also elaborates on questions you may ask.
2. Consult with NHMRC
Early conversations with NHMRC can help developers decide whether or not to proceed with development of this particular guideline. Guideline developers seeking NHMRC third-party guideline approval should discuss with NHMRC the need for guidelines in their chosen area, even at the conceptual stage, as there may be current guideline activity of which you are unaware that overlaps with or affects your guideline.
Other considerations:
- NHMRC can provide advice about whether your guideline will address current government priorities.
- NHMRC procedures for third party guideline approval have set milestones and deadlines that must be met in order for the approval process to proceed. If you intend to seek NHMRC approval you will need to get an idea of what is expected and when — it will determine whether your guideline can be accomplished within the required timeframe (see Procedures and Requirements ).
If you decide to proceed with a clinical practice guideline you should register your intent on the Australian Clinical Guidelines Registration Page. Public and environmental health guidelines do not have such a requirement at this stage.
3. Source your funding
Guidelines can be very expensive depending on their size and scope (see Section 7). If your organisation or commissioning body is unable to fund your guideline entirely there are a number of ways you can potentially source funding. These can include:
- granting bodies
- charitable foundations
- institutions, such as universities, research institutes or colleges
- Commonwealth and state/territory health departments and other health agencies
- other government agencies that may have an interest in your guideline, such as the Department of Defence, Comcare, National Disability Insurance Scheme, Department of Veterans Affairs, the Department of Agriculture and Water Resources
- not-for-profit groups such as health advocacy foundations and charities
- international funding bodies and collaborations
- crowd-funding.
Partnering with other groups around the country to form national collaborations is not only effective for streamlining resources, it can also broaden stakeholder networks and boost the credibility of your guideline by ensuring that consistent advice is provided across the country. For example, NeuRa and Kidsafe jointly funded the National Guidelines for Safe Restraint of Children Travelling in Motor Vehicles. Consider whether your guideline can be included under the umbrella of broader topic areas to increase opportunities for funding from organisations with an interest in these areas — for example, men’s health or cardiovascular disease. Organisations that have previously only funded research activities may also be interested in funding guidelines that translate this research into best practice.
Be creative about the possible ways that you can fund your guideline or obtain resources that can cut down costs. Some organisations may not directly fund your guideline but may support you by providing project staff and other useful resources. They may also fund the evidence review of a discrete topic of interest that is within the scope of the guideline. For example, the Australian Radiation Protection and Nuclear Safety Agency conducted a review on radiological quality of drinking water and this was used to update a chapter of NHMRC’s Australian Drinking Water Guidelines.
Be aware that depending on the topic, groups with vested interests may be interested in providing funding for your guideline. It is important to conduct due diligence on potential funders as undue influence from organisations funding and sponsoring guidelines can be a potential contributor to bias in guideline development (IOM 2009; IOM 2011). It may then be helpful to consider:
- Will your source funding affect the trustworthiness of your guidelines? For example, WHO guidelines may not be funded by commercial entities (WHO 2014).
- How much involvement or influence will the funding or commissioning organisations have or expect to have in the guideline development?
- Will you need to put a policy in place to manage conflicts of interest and other risks of bias associated with the funding organisations?
- Will you need to report back about how funds were spent?
For the purposes of transparency all sources of funding will need to be declared in your final guideline even if crowdfunding has been used as your funding source.
For agencies that fund and produce their own guidelines this would include providing a statement to detail what the funding was used for. An example is the National Blood Authority’s (NBA’s) Patient Blood Management guidelines, which state that ‘the NBA provided the secretariat, project funding and project management.’
If you are receiving funds from external sources, you will also need to establish reporting requirements for each funding body before you start development and include this in your project plan. Strategies should be put in place to manage and document their involvement in the development process, as including the funding or commissioning body in the development process, even as an observer, may be perceived as a potential conflict of interest. This information is reviewed when assessing the quality of a guideline (see the AGREE II tool).
You may consider including representatives from the funding or commissioning body in an oversight committee — this will ensure that they are not directly involved in the development process, processing of the evidence or formulating recommendations. You should also aim to include independent observers wherever possible to ensure that there is an oversight process, particularly with guideline funding arrangements involving tax payer funds (see the Identifying and managing conflicts of interest module).
4. Establish who will be involved and when
Governance of your guideline project is an important issue as it determines who makes decisions and when. Many different people will be involved in the development of your guideline, each with different roles and responsibilities and different times when they will be required or consulted. You should document in your project plan when you need to involve individuals or groups and what you will be asking them to do. You may also need to allocate resources to recruit them and pay for their time (see Table 1 in Section 7). For example, organisation and planning may be undertaken by the project management team, the chair of your guideline development group or some independent experts to help plan the process — this may depend on your organisational procedures and available resources.
Decisions about the guideline recommendations will be made by the guideline development group with input from advisory committees or experts.
Processes will need to be put in place to record details about these stakeholders including their roles, experience and expertise, meeting attendance and disclosed interests. You will need to develop a process to keep this information up to date for transparency. You should also document each stakeholder’s input into the guideline as much as possible for tracking purposes and for acknowledging contributions in the final guideline.
Advice on how to establish the guideline development group, engage stakeholders, involve consumers, find independent reviewers and conduct public consultation is provided elsewhere on the Guidelines for Guidelines site.
5. Develop a project plan
A project plan will need to be developed by the guideline project manager. The plan should include details on:
- the rationale for and scope of the guideline
- the intended end users and target populations
- the roles and responsibilities of those involved in project governance, for example, terms of reference, a code of conduct or confidentiality agreements
- project timelines, including deadlines for meeting project milestones, planned meetings, reporting requirements and downtime waiting for other organisations — the KHA-CARI timeline template is a good example for planning project timelines
- potential procurement activities, including contracts and agreements
- a risk assessment and management plan to accommodate contingencies like loss of key staff, not meeting deadlines, data and file security, running out of funds and scope creep
- a protocol for all the steps in the guideline development process — this can be completed and updated as the project progresses and circulated in the development group so that everyone can see that the project is on track
- a description of the tools and platforms used for collaboration and development
- a budget for guideline development (see Section 7).
A Gantt chart is a good way to view and track timelines for the whole guideline development process. It can be a useful single point of reference for all parties (see Figure 1, underneath). This template is available to download as an attachment to this page.
Figure 1: Example Gantt chart for project planning
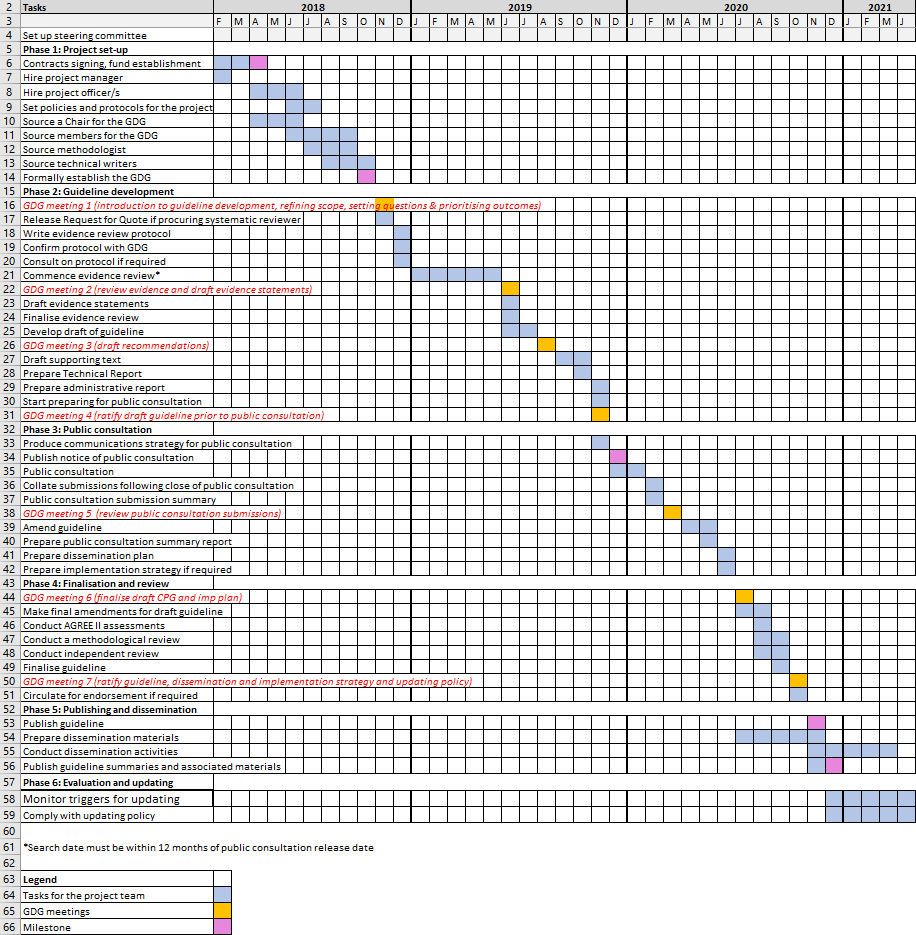
6. Consider other issues
Before you start you should consider other issues that may affect guideline development processes and your planned use of resources such as:
- Can the guideline topic be broken up into different projects that can be tackled individually?
- Are there already activities underway or evidence reviews in development that can be referred to or partnered with to minimise resource use and avoid duplication of effort?
- Are there any requirements attached to project funding, such as penalty clauses or sign-off by organisational bodies?
- Are there any legal issues such as copyright, privacy considerations or data use that require specialist legal advice?
- Is the guideline topic contentious — does a communications strategy need to be in place to deal with public interest during development? If so, who is the spokesperson for the guideline development group?
- Do acknowledgement policies for authors, contributors and editor need to be put in place?
- Do resources need to be allocated to lodge your evidence records in data repositories?
7. Plan your budget
Guideline development can be an expensive process and can easily run over budget if not planned and funds allocated carefully. Guideline development should not be attempted without sufficient funding. There is no set formula for how to budget for a guideline; however, it is important to try and capture known essential costs to cover staffing and meetings, and to estimate what will be required for the more complex activities such as the evidence review. Table 1 (immediately below) is an example of the resources that may be required and what should be budgeted for.
| Item | Details | Budget |
|---|---|---|
| People | ||
Project staff Project manager, project support officers, executive or leadership team
| A guideline will require a project manager to manage the guideline development group, coordinate meetings and run consultation activities. The executive or leadership team will need to commit resources to activities, promote the project, establish partnerships and collaboration opportunities. | Salaries for staff and input as required from executive or the leadership team. This is to be maintained for up to 2 years depending on the scope and complexity of the guideline. |
| Steering committee | A small group of people to help set up the process, including setting the scope and nominating individuals for the guideline development group. | Costs may include a face to face meeting. |
| Guideline development group | For large guidelines you may need 6–8 meetings, with the understanding that the guideline development group will need time to prepare for meetings. Consider if training is necessary and how that can be delivered, e.g. introduction to the guideline process, how to interpret levels of evidence and to formulate recommendations based on evidence, cultural awareness training. | Costs will differ depending on the size of your committee, where they live, whether the preference for meetings is face to face or via teleconference or videoconference. Venue and travel costs will need to be accounted for. Training may involve attendance of a facilitator at a meeting. Budget for their fee for service and travel costs. |
| Stakeholder engagement | There are a variety of methods that can be used to engage stakeholders (See Engaging stakeholders). | Some methods will be more costly than others. Consider which methods will be most important for your guideline and budget appropriately. |
| User and/or consumer testing | Depending on the method (see Engaging stakeholders). Costs associated with setting up focus groups (including a facilitator, venue and travel costs). | NHS INVOLVE program in the UK has an example budget calculator to assist in budgeting (in GBP).
|
| Products or activities | ||
| Development software | There are a number of guideline development platforms, e.g. MagicApp, GRADEpro GDT, or systematic review software, e.g Covidence, DistillerSR that can be used. You should choose a platform that is appropriate for the size of your guideline. | Some software is freely available to use but others such as MagicApp, or GRADEpro GDT will need a license negotiated with the software developers. |
| Publishing | There may be many formats to budget for. Also consider that some hosting platforms will cost money. | Costs will vary depending on the format the guideline is published in. |
| Derivative products | This could include paying for any translation activities into different languages, developing decision aids or pamphlets. Also could include development of phone apps. | Translation services should be sourced from an NAATI accredited provider. |
| Dissemination and implementation activities | Conferences, champions/advocate speaking appearances. Communications or media experts could also be required. | |
| Sponsorship or endorsement from other bodies | Some colleges require payment to endorse specific guidelines. | |
| Additional expertise (consider contracting out) | ||
Evidence review
| The evidence review process should be undertaken by qualified people with experience in this work.
Experienced people may already be involved in the project, but could otherwise be acquired through additional recruitment or by contract. The final deliverable will be the systematic review and/or a literature review being presented as a separate technical report. A statistician or librarian may also need to be sourced to assist with this work.
You may also need to budget for licenses for software to support the systematic review process, for example, Covidence.
| Depending on the scope of your guideline and number of questions, costs associated with the evidence review can vary considerably. More funds will need to be allocated if:
Given the skill required to undertake the evidence review, it is important that there is a quality assurance process in place to check the results of the review before the guideline is publicly released. This might require budgeting for extra personnel, for example, a methodologist who is independent of the process. |
| Methodologist(s) | They can help guide the guideline development group through the process in a contracted role, or perhaps as a member of the group. Methodologists can help draft, advise on and/or undertake the search strategy, appraise the evidence and develop evidence tables. | Budget for the methodologist to attend all meetings where there is a discussion of the evidence and forming of recommendations. |
| Technical writing | Technical writers will be responsible for producing multiple drafts of the guideline and perhaps additional reports — public consultation report or administrative/process report. | Budget for technical writer/s to attend all meetings. You need to also factor in whether they will need to produce associated reports such as a public consultation report. |
| Copy-editing | Proofreading, desk-topping and editing for the draft prior to public consultation or for the final draft. | This is different to contracting a technical writer. A copy-editor will edit the full guideline in accordance with a Style manual and also check for any inconsistencies. |
8. Review plan and monitor progress
Your project plan and budget need to be examined together to ensure that the project is feasible. It should also be constantly monitored during guideline development.
Ask yourself:
- Do you have enough funds to complete your guideline?
- Where funding has been allocated are the timelines, funding levels and deliverables for the project realistic?
- Do they need to be adjusted to make the guideline more feasible?
- Do you have plans in place for any setbacks identified in your risk assessment such as scope creep?
- Have you put policies in place that will minimise risk of bias?
If you need to make any changes to your project plan, remember to keep track of these changes using strict version control and update anyone who needs to know. Version control and good documentation will also help with long-term planning for updates and evaluation of the project. This would be useful if you are reflecting on the ‘lessons learnt’ from the project by assessing the variances from the original timelines and budget and the reasons why these occurred.
9. Hire contractors
You may need to seek specialist expertise to undertake guideline tasks such as the evidence review or technical writing. If you will be contracting out services it is important to adhere to your organisation’s procurement policy in order to ensure probity of process and demonstrate value for money. It is also important to establish and maintain a clear audit trail and comply with any organisational policies or legislative obligations.
Government bodies have access to panel arrangements for services that may be routinely required by that agency. This includes technical writers, economists, methodologists, systematic reviewers and editors. If you are being funded by a government agency, check whether you may have access to those panel arrangements.
10. Contingency planning
Despite careful planning, there may be reasons outside of your control for why a project may run over time or budget. Ensure you refer to these in the risk framework in your project plan, such as evidence reviews taking longer than thought due to an unexpected larger body of literature; staffing issues requiring replacement of, or adding more, staff to your team; or an inability to find, or delay in finding experts for particular tasks.
You need to make provisions for retaining key staff to complete the guideline if there are unexpected delays. Regular progress reports or meetings with your steering group, funder or executive should be part of a risk mitigation strategy so they are aware of and prepared for issues if things are not going to plan.
Useful resources
Developing NICE guidelines: the manual
SIGN 50 – A guideline developer’s handbook
G-I-N McMaster Guideline Checklist
WHO Handbook for Guideline Development (Second Edition)
NSW Health Public Health guidance series
References
National Health and Medical Research Council (NHMRC) (2014). Australian Clinical Practice Guidelines 2014 Annual Report.
Acknowledgements
Thanks to Dr. Philip Alderson from National Institute for Health and Clinical Excellence (NICE), UK for his advice on developing this module.
Version 5.0. Last updated 5 September 2019.
Suggested citation: NHMRC. Guidelines for Guidelines: Project planning. Last published 5 September 2019
Changes to this module
February 2026
*An older version of this module developed between 2018-19 indicated that guidelines cost approximately AUD $1 million. This was based on data NHMRC had collected on externally developed guidelines outlined in the Annual Report on guidelines (NHMRC 2014). From feedback reported to us by guideline developers, the cost of national guideline development is now closer to AUD $3 million depending on the scope. NHMRC asks that funding amounts be specified in the Administrative Report published alongside the guideline for transparency. This information will assist the guideline development community in future scoping, project planning and management endeavours.