The Embryo Research Licensing Committee (ERLC) of the National Health and Medical Research Council has considered whether specific embryo models fall within the regulation of the Research Involving Human Embryos Act 2002 and Prohibition of Human Cloning for Reproduction Act 2002. The information below is based on ERLC’s understanding of the science at the time it was considered.
Researchers are encouraged to contact ERLC before commencing research using human embryo models in Australia. The key considerations for ERLC were whether the entity meets the definition of a human embryo as described in the legislation, and the intent of the ‘14-day rule’.
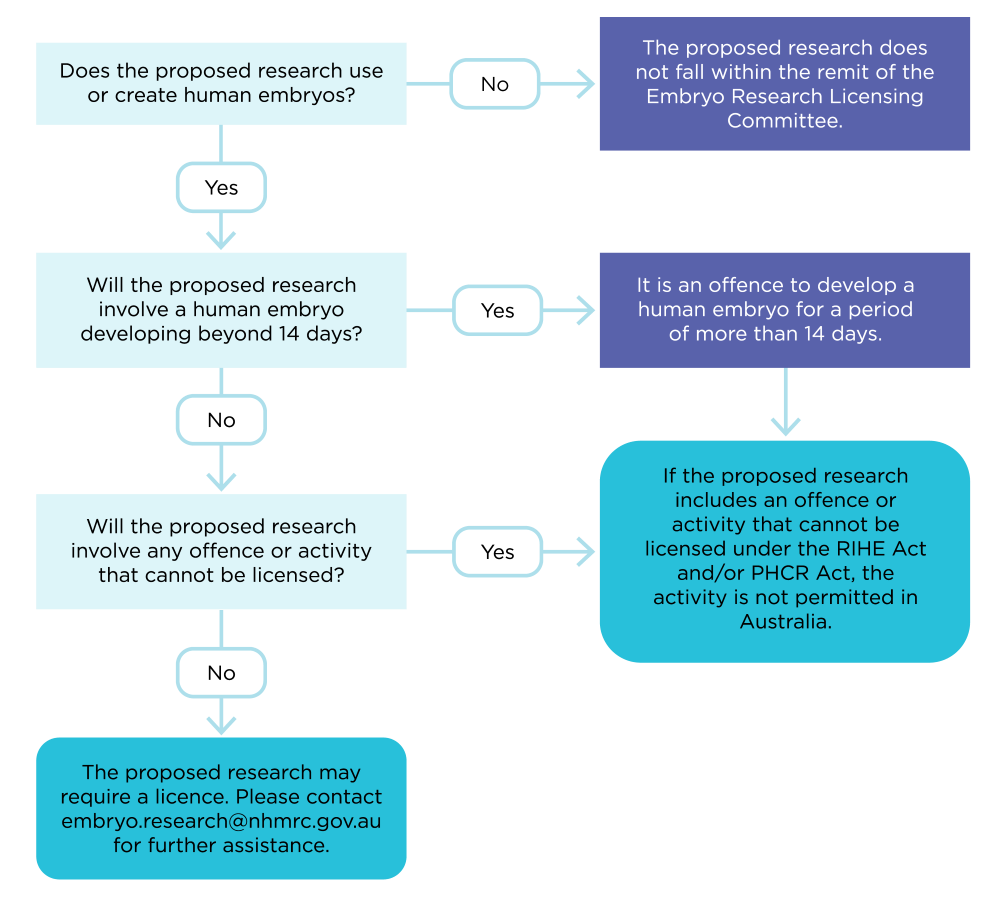
Decision flowchart
This flowchart has been developed to assist researchers in Australia to determine whether a specific embryo model may be regulated by the Research Involving Human Embryos Act 2002 (RIHE Act) and Prohibition of Human Cloning for Reproduction Act 2002 (PHCR Act). For each question, refer to the supporting information to assist in the assessment. The information in this flowchart and supporting information reflects the science at the time it was developed, as it was understood by ERLC. If there is any doubt about whether an embryo model is regulated, please contact ERLC at embryo.research@nhmrc.gov.au.
Figure 1. Decision Flowchart. Refer to the Supporting Information section below to answer the questions in the flowchart.
Supporting Information for the Decision Flowchart
Does the proposed research use or create human embryos?
An entity is considered as a human embryo if it fulfills all the requirements below:
- The entity is human cells with a human nuclear genome or altered human nuclear genome (including human stem cells, induced pluripotent stem cells, somatic cells, gametes, etc); and
- The entity is discrete, capable of being identified or counted; and
- The entity has the potential to develop up to, or beyond, the stage at which the primitive streak appears. An embryo model meets this definition even if it does not: follow the normal embryonic development process, or undergo epithelial-to-mesenchymal transition (EMT) and/or form a primitive streak, or pass through the stage of gastrulation; and
- The entity does not show morphological features of an embryo after 8 weeks development (e.g. organs, limbs, heartbeat); and
- The entity has arisen from fertilisation or any other process that initiates organised development of a biological entity. It has met this requirement if:
- There is evidence of developmental attributes contributing to spatial and/or temporal organisation within the entity; and
- It is an intact conceptus, which has the potential to develop the full complement of parts of a normally developing embryo (including embryonic cells that give rise to all the cells in a human body and extraembryonic tissues such as the amnion and placenta).
The entity is not required to exist independently to fulfil this requirement.
If all the above requirements are met, your entity is considered a human embryo.
You also need to consider if the creation of the entity involves the use of a human gamete (egg or sperm), human embryo or excess ART embryo.
This includes, but is not limited to:
- use of a human embryo to create of human embryonic stem cells
- fertilisation of a human egg.
If it does, your research uses human embryos.
Will the proposed research involve the development of a human embryo beyond 14 days
Development of a human embryo for a period of more than 14 days, excluding any period when development is suspended, is not allowed.
During the course of development of ‘other embryos’, created by a process other than fertilisation, the ‘other embryos’ may not go through the same linear stages of embryonic development in the same timeframe as embryos created by fertilisation. Because of the difficulty in applying the so-called ‘14-day rule’ to these ‘other embryos’, they are only allowed to develop to morphological and/or molecular stages equivalent to an embryo at 14 days post fertilisation.
Does the proposed research involve any offences as set out in the RIHE Act or PHCR Act?
This includes, but is not limited to:
- creation of a chimeric embryo
- creation or use of a hybrid embryo
- giving or receiving valuable consideration for the supply of a human egg or embryo.
Refer to sections 10 to 12 of the RIHE Act and sections 9 to 23B of the PHCR Act for offences under each Act.
Application of the legislation to specific models
ERLC has applied relevant Australian legislation to determine whether the specific embryo models detailed below met the definition of a human embryo and could consequently be regulated by the licensing scheme described in the RIHE Act. ERLC’s determinations apply only to the specific model considered and ERLC’s understanding of the science at the time it was considered.
Models of an early human embryo (for example, blastoid)
In 2021–22, ERLC considered an embryo model of the human blastocyst, an early stage of human embryo development, referred to as a blastoid. Researchers sought ERLC's advice when studies reprogramming fibroblasts led to the development of a blastoid.
The key features of the blastoid model at the time it was considered were an inner cell mass-like structure containing epiblast and primitive endoderm-like cells, a blastocoel like cavity and a trophectoderm-like outer layer of cells.1
ERLC determined that this blastoid model met the definition of a human embryo as described in the RIHE Act. Creation of this model may be permitted under a licence approved by ERLC. Blastoids created under licence are not permitted to develop beyond a morphological or molecular stage equivalent to a normally developing human embryo 14 days post fertilisation.
Models of a human embryo around the time of gastrulation (for example, Moris 3D gastruloid model)
In 2023, ERLC considered a 3D gastruloid model, an in vitro structure that resembles the human embryo in the immediate post-implantation stage of development. Researchers sought ERLC’s advice on the creation of this model using human embryonic stem cells, leading to a 3D structure with three germ layers, but without additional extra-embryonic tissues, as described in Moris et al2. This model is referred to here as the Moris 3D gastruloid model.
ERLC found that the Moris 3D gastruloid model met some parts of the definition of a human embryo. However, ERLC found that the model did not fully demonstrate “organised development of a biological entity” on the basis that it is not an intact conceptus with the potential to develop the full complement of parts of a normally developing embryo (including embryonic cells that give rise to all the cells in a human body and extraembryonic tissues such as the amnion and placenta) as it does not have all extraembryonic cell lineages.
ERLC concluded that the Moris 3D gastruloid model does not meet the definition of a human embryo under the RIHE Act and therefore does not require an embryo research licence to be granted for creation or use in research in Australia at this time.
ERLC noted that a licence would be required if a researcher planned to create their own embryonic stem cell lines to use as the source of cells for the Moris 3D gastruloid model. ERLC also noted that if the source is commercially available established embryonic cell lines, use and approval of the cell lines is regulated through the National Statement on Ethical Conduct in Human Research (2023) via the institution’s Human Research Ethics Committee.
ERLC noted that there is potential for some other 3D gastruloid models to satisfy all the requirements to meet the definition of a human embryo, for example, if the entity was an intact conceptus. As such, researchers are encouraged to consider the attributes of each specific model before commencing research and to seek advice from ERLC.
Notes on ERLC’s interpretation of the legislation
ERLC recognises that the object of the RIHE Act is to address concerns, including ethical concerns, about scientific developments in relation to human reproduction and the utilisation of human embryos by regulating activities that involve the use of certain human embryos created by assisted reproductive technology or by other means.
ERLC has applied a broad interpretation of the meaning of “organised development” in line with its understanding of the intent of the legislation. The interpretation is applied on the basis of current understanding of this emerging field of science. This broad interpretation supports the public good by allowing certain types of research to occur where the full complement of cell types required to form an embryo are not present. Of particular importance is ensuring that no prospect of sentience exists.
The ‘14-day rule’ is a legal and regulatory timeframe that limits in vitro human embryo research to the period before the primitive streak appears. The primitive streak is a transient structure whose formation, on day 15 of human embryo development, marks the start of gastrulation, the process in which the inner cell mass is converted into the trilaminar embryonic disc, which is comprised of the three germ layers (ectoderm, mesoderm and endoderm).
ERLC’s determination of the 14-day milestone is not by calendar day but by the morphological and molecular features equivalent to the gastrulation stage. This aligns with both scientific and legal approaches to the rule and takes into account ERLC’s view that the 14-day timeline was intended to mark the time when gastrulation occurred in the normal embryonic development process. This interpretation is based on ERLC’s best current scientific understanding of embryo models, including those that do not go through the linear stages of normal embryonic development.
Resources
- Prohibition of Human Cloning for Reproduction Act 2002
- Research Involving Human Embryos Act 2002
- Research Involving Human Embryos Regulations 2017
- Customs (Prohibited Import) Regulations 1956 (section 5L)
- Customs (Prohibited Export) Regulations 1958 (section 8A)
- NHMRC website - Import and Export of Cell Lines from Human Embryo Clones
- Ethical guidelines on the use of assisted reproductive technology in clinical practice and research 2007 (updated 2023)
- National Statement on Ethical Conduct in Human Research 2023 (effective 1 January 2024)
- National Statement on Ethical Conduct in Human Research 2007 (current to 31 December 2023)
1 Liu, X., Tan, J.P., Schröder, J. et al. Modelling human blastocysts by reprogramming fibroblasts into iBlastoids. Nature 591, 627–632 (2021). https://doi.org/10.1038/s41586-021-03372-y
2 As described in: Moris, N., Anlas, K., van den Brink, S.C. et al. An in vitro model of early anteroposterior organization during human development. Nature 582, 410–415 (2020). https://doi.org/10.1038/s41586-020-2383-9.