The ability to make insulin was one of the great medical breakthroughs of the 20th Century. However, it quickly became clear to clinicians and researchers how complicated the human body's systems were for regulating and using blood sugar. In the absence of a deep understanding of these systems, providing insulin could have dangers.
Supported by funding from the National Health and Medical Research Council (NHMRC), researchers at the Garvan Institute of Medical Research made significant contributions to knowledge about insulin delivery and fundamentally changed and improved clinical practice worldwide.
A PDF version of this case study is available in the Downloads section below.
Origin
Before mass-production of insulin commenced in 1923, for those (usually young) people who developed what we now call type 1 diabetes, 'life was miserable, and usually very short'.1 They would probably die from a condition called diabetic ketoacidosis (DKA), also known as 'diabetic coma'.2
Injectable insulin was literally a lifesaver. It could be used to treat DKA and to prevent the damaging effects of diabetic hyperglycaemia: the high blood sugar [blood glucose, BG] levels experienced by people with both type 1 and type 2 diabetes.
However, being able to provide insulin was not the same as knowing how best to provide it. By the 1960s, longer and shorter acting insulins had been developed for subcutaneous bolus (single, large dose) injection to enable diabetic patients to improve BG control with and between meals, but this was a poor approximation of the way that the body produced its own insulin.
Moreover, for DKA treatment there was significant variation internationally in how much insulin was being given and whether it was being injected into a vein, under the skin or into a muscle.3,4 Generally though, the amounts injected tended to be large – up to 500 units at a time – leading to unpredictable and often harmful effects on BG and blood potassium levels.
The lack of an evidence-based approach combined with a 'more is better' philosophy of insulin injection was confusing for doctors and dangerous for patients, who continued to die from DKA despite the availability of insulin.
Hospital staff, who were on the 'front line' when treating DKA, needed more knowledge, and people living with type 1 diabetes needed an improved ability to manage their insulin levels throughout the day, if they were to prevent DKA emergencies from occurring in the first place.
Investment
In 1963, when St Vincent's Hospital Sydney established a new research department (now the Garvan Institute of Medical Research), improving understanding of when and how to provide insulin to diabetic patients was a key concern for the institute's director, Dr Leslie Lazarus.
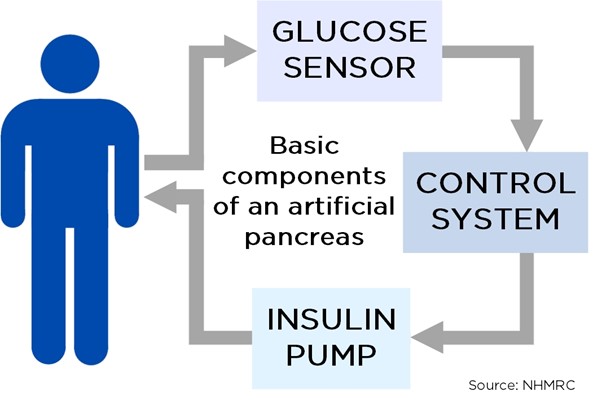
In 1970, Ted Kraegen completed his PhD research at the institute. His project aimed to measure the magnitude and delay times of changes to BG levels in response to small infusions of insulin. The ultimate goal of this research was to contribute to the development of a 'closed loop' BG feedback control system. Also known as an 'artificial pancreas', such a system would measure a patient's BG levels and, informed by a control system, adjust insulin delivery accordingly in real-time to keep BG within safe parameters.
Kraegen commenced work at the institute as an NHMRC Senior Research Officer, employed on an NHMRC Project Grant held by Lazarus. Supported by further NHMRC grants plus funding from the Ramaciotti and Kellion Foundations, a team of Garvan Institute researchers including Lazarus, Kraegen, Lesley Campbell and Don Chisholm, worked to make the artificial pancreas a reality.
Research
The Garvan Institute team created electronic systems for continuous BG monitoring. These systems, based upon modifications to off-the-shelf components, received a flow of blood from test subjects via a catheter, then divided this flow into discrete segments that could be continuously tested for their BG levels.
These systems generated sensitive plots of BG responses to insulin infusions which showed that infusions of only 2.4 units of insulin per hour were sufficient to reduce BG levels continually in both healthy and diabetic subjects. In fact, a 30-minute-long infusion of one unit of insulin produced a greater reduction in BG levels than a single intravenous injection of the same quantity.
These findings made sense because the 'half-life' of insulin when injected intravenously was known to be very short: only 3 to 5 minutes. Consequently, most of the insulin provided through a bolus intravenous injection might often be wasted.
Realising the significance of their findings for DKA treatment, Lazarus decided to trial a low-dose approach in a hospital setting. Working with clinicians John Casey and Warren Kidson, Kraegen tested the new approach in 11 patients who presented to St Vincent's Hospital with symptoms of DKA or hyperglycaemia. They found that rates of intravenous insulin infusion of 1.2 or 2.4 units per hour were sufficient to return insulin levels to those of non-diabetic subjects. Published in 1974, the Garvan Institute team's proposal for low-dose insulin infusion led to rapid international acceptance and widespread uptake.5
In 1975, the Garvan Institute team completed the first version of the artificial pancreas, a system that took up half a room. This device, a forerunner of a continuous subcutaneous insulin infusion (CSII) system, was one of only three in the world.
Results
The Garvan Institute team made several associated breakthroughs in which Campbell played a key role.
They realised that, because insulin tends to stick to the infusion apparatus, up to 80% of added insulin could be lost when using low doses, creating the danger that insufficient insulin would reach the patient. They then tested ways of making the insulin less 'sticky' and recommended a procedure that became part of standard clinical practice.
In addition, they demonstrated that type 1 diabetic patients, during a low-dose insulin infusion, were unable to resist falls of BG by releasing insulin counter-regulatory hormones (including glucagon, epinephrine, cortisol and growth hormone) that increase BG. This first recognition of the 'counter-regulatory defect' as a cause of susceptibility to hypoglycemia (low BG) was also important in efforts to improve stability of BG control.
Regarding the daily living of people with diabetes, they found that when insulin is infused subcutaneously its effects occur more slowly and last much longer. In fact, the BG response to changes in the rate of subcutaneous infusion may take two hours or more.
This finding had a significant implication for the design of CSII systems as it meant that there was no point making frequent adjustments to the subcutaneous infusion rate and that any adjustments had to be made hours before the time of the intended effect.
Another major finding was that provision of insulin before a meal was more effective in controlling BG levels than providing it in response to BG rises after a meal. This observation meant that CSII systems should be 'anticipatory' and 'open loop' for meals and that, ideally, diabetic patients would be able to inform a CSII that they were about to eat, and how many calories they were about to consume.
Outcomes and impact
The low-dose insulin infusion approach for treating DKA and hyperglycaemia pioneered by researchers at the Garvan Institute remains the default approach worldwide and informs hospital practice in Australia every day.
In 2014–15, there were 7,132 hospitalisations in Australia with a principal diagnosis of DKA. Most of these hospitalisations (5996 or 84%) were for people with type 1 diabetes, and of these, half (3245 or 54%) were for children and young people aged under 25.6 However, in 2014 there were only 3 deaths in Australia from diabetes among those under 25.7 This contrasts with mortality rates often around 12% before the institute's DKA work.
Development of a safe and effective CSII system remains a key goal of diabetes researchers internationally and significant progress has been made. In 2016, the US Food and Drug Administration (FDA) approved a device8 that measures BG levels every 5 minutes and provides small amounts of insulin, responsive to BG, throughout the day to mimic the background insulin production of the pancreas.
It can also deliver larger amounts of insulin ahead of meals, just as the Garvan Institute researchers suggested.
Timeline
| Year | Activity |
|---|---|
| 1968 | Grants (Growth hormone studies) |
| 1970 | Equipment Grant (Garvan) Grant (Insulin secretion) |
| 1973 | Grant (Insulin secretion) |
| 1974 | Low-dose insulin infusion reported Grant (Biological control systems |
| 1975 | Artificial pancreas developed |
| 1977 | Grants (Biological control systems) |
| 1979 | Grant (Diabetic complications), Grants (Computer-assisted infusion system) |
Researcher profiles
Professor Leslie Lazarus AO
Leslie (Les) Lazarus graduated in medicine in 1953 and in 1962 became the first full-time specialist in endocrinology at St Vincent's Hospital Sydney. In 1963, Lazarus was one of the initial co-directors of the Garvan Institute and became the first sole Director (1969–1990). He was Associate Professor and then Professor of Medicine at the University of New South Wales (UNSW) (1974–1995). Lazarus was continuously involved with the Council of the Endocrine Society of Australia (1968–1982) and was an elected Honorary Life Member. In 1988, Lazarus was made an Officer of the Order of Australia.
Professor Edward Kraegen AO
Edward (Ted) Kraegen became UNSW's first biophysics honours graduate in 1965. He joined the Garvan Institute in 1967 as a PhD student and in 1973 received a Winston Churchill Fellowship.
Kraegen became head of the Garvan Institute's Diabetes Research Group in 1990 and was Associate Professor and then Professor of Medicine at UNSW (1987–2009). In 2006, he was made a Life Member of the Australian Diabetes Society and in 2007 received a Citation for Distinguished Service from the American Physiological Society. Kraegen was made an Officer of the Order of Australia in 2019.
Professor Lesley Campbell AM
Lesley Veronica Campbell graduated in medicine in 1968. She joined the Garvan Institute in 1979 as a principal researcher and is currently an Emeritus Principal Research Fellow in the Clinical Diabetes, Appetite and Metabolism Lab.
Campbell is Professor of Medicine, St Vincent's Clinical School, Faculty of Medicine, UNSW Sydney and was Director of the Diabetes Centre at St Vincent's Hospital Sydney (1990–2014). She received the medal of the Swedish Society of Medicine in 2001, was made a Member of the Order of Australia in 2008 and received the Australian Diabetes Society Lifetime Achievement Award in 2020.
Professor Don Chisholm AO
Don Chisholm was head of the Garvan Institute's Diabetes and Metabolism Research Program (1978–2003) and Foundation Director of the Diabetes Centre at St Vincent's Hospital Sydney (1980–1991). Chisholm was made an Officer of the Order of Australia in 1999.
Drs Kidson and Casey
Warren Kidson was a consulting endocrinologist at the Prince of Wales and Sydney Children's Hospitals. John Casey was a consulting endocrinologist at St Vincent's Hospital Sydney
Diabetic ketoacidosis
Insulin normally plays a critical role in allowing sugar (glucose) – a major source of energy for muscles and other tissues – to enter cells. When insulin is in short supply the body can instead begin to break down fat for energy. This process can lead to a dangerous build-up of ketones (acidic molecules) in the bloodstream which, if left untreated, can lead to diabetic ketoacidosis (DKA). DKA is sometimes the first sign of type 1 diabetes in people who have not yet been diagnosed. It can also occur in someone who has already been diagnosed with diabetes but who has an infection, illness or injury, or who has missed doses of insulin or otherwise been unable to obtain it.
Continuous blood glucose monitoring
A major impediment to the development of a CSII for diabetic patients to wear and use throughout the day has been the lack of a stable and portable glucose sensing mechanism. When continuous BG monitoring first became commercially available in 2000 its measurement error was more than +/- 20%. However, since that time the accuracy of such units has significantly improved, their size, weight, complexity and cost have decreased and their duration of use, user-friendliness, user interface and displays, data management and software for data analysis have all improved. Ongoing advances in technology and clinical research are expected to improve these units still further.9
References
This case study was developed with input from Professor Ted Kraegen, Professor Don Chisholm and Medtronic Australasia Pty Ltd and in partnership with the Garvan Institute of Medical Research.
The information and images from which NHMRC Impact Case Studies are produced may be obtained from a number of sources including our case study partner, NHMRC's internal records and publicly available materials.
The following sources were consulted for this case study:
- Dhatariya K. The evolution of DKA management. The evolution of DKA management. Br J Diabetes Vasc Dis 2015;15:31–33
- Tattersall R. Diabetic ketoacidosis 1850–1970. Tattersall's Tales. Diabetes Digest 2012;11(1):8–9
- Kidson W, Casey J, Kraegen E, Lazarus L. Treatment of severe diabetes mellitus by insulin infusion. Br Med J. 1974 Jun 29;2(5921):691
- Tattersall R. Diabetic ketoacidosis 1950 onwards. Tattersall's Tales. Diabetes Digest 2012;11(2):68–70
- Madison LL. Low-dose insulin: A plea for caution. New England Journal of Medicine. 1976 Feb 12;294(7):393–4
- Australian Institute of Health and Welfare 2016. Diabetic ketoacidosis (DKA) among children and young people with type 1 diabetes. Diabetes series no. 26. Cat. no. CVD 77. Canberra: AIHW
- Australian Institute of Health and Welfare 2021. General Record of Incidence of Mortality (GRIM) books. https://www.aihw.gov.au/reports/life-expectancy-deaths/grim-books/contents/grim-excel-workbooks. AIHW Website, accessed 22 March 2022
- FDA approves first automated insulin delivery device for type 1 diabetes [Internet]. U.S. Food and Drug Administration. 2022 [cited 25 February 2022]. Available from: https://www.fda.gov/news-events/press-announcements/fda-approves-first-automated-insulin-delivery-device-type-1-diabetes
- Rodbard D. Continuous glucose monitoring: a review of successes, challenges, and opportunities. Diabetes technology & therapeutics. 2016 Feb 1;18(S2):S2-3
Partners
